Have you ever stared up into the night sky during a dazzling fireworks show and wondered exactly is magnesium used in fireworks?
At Red Apple Fireworks, we're excited to confirm that yes—magnesium is an essential ingredient! Magnesium adds incredible brightness and sparkle to fireworks, making your celebrations memorable.
From DIY backyard shows to professional-grade pyrotechnics, magnesium plays a key role in creating unforgettable displays.
Let’s dive deeper into this exciting part of the chemistry of fireworks and explore what makes magnesium so important.
What this article covers:
- How Magnesium Creates Bright White Sparks
- The Bright White Light of Magnesium
- Other Uses of Magnesium in Pyrotechnics
- Safety Considerations of Magnesium in Fireworks
How Magnesium Creates Bright White Sparks
So, is magnesium used in fireworks? Absolutely—and it’s one of the most important ingredients for creating brilliant, high-intensity light.
Magnesium is prized for its ability to burn at extremely high temperatures—around 5,610°F (3,100°C)—producing a pure, white-hot glow that no other element can match.
When magnesium ignites, it burns rapidly and brightly, filling the sky with intense white sparks.
This makes it perfect for adding contrast and clarity to firework displays, especially during grand finales and high-altitude bursts.
It also enhances the vibrancy of coloured effects by acting as a white-light amplifier, making reds redder, blues bolder, and greens more vivid.
Whether it’s in professional-grade aerial shells or compact cakes and rockets, magnesium ensures that your display isn’t just seen—it’s remembered.
From huge events to backyard celebrations, magnesium brings the brilliance that turns good fireworks into spectacular ones.
How Magnesium Enhances Firework Colors
Magnesium isn't responsible for bright reds or blues—that's where metallic salts like strontium and copper come into play.
But magnesium is irreplaceable when it comes to enhancing brightness and brilliance. It makes colors more vibrant, sharp, and visible, especially in large aerial shells and intricate displays.

The Bright White Light of Magnesium
Now back to magnesium—the star behind those dazzling white bursts lighting up the night sky.
How Magnesium Burns in Fireworks
When ignited, magnesium burns at extremely high temperatures, around 5,610°F (3,100°C). This intense heat and energy create a brilliant white glow unmatched by other materials.
Magnesium ignites rapidly, making it ideal for immediate, spectacular effects that catch everyone's eye.
Why Magnesium Is Essential for Intensity and Visibility
Magnesium is the go-to ingredient for creating eye-catching brilliance in fireworks. Its intense white light enhances visibility, contrast, and overall impact—especially during large-scale events or grand finales.
Because it burns at extremely high temperatures, magnesium helps every color in a fireworks display pop, amplifying reds, blues, and greens by acting as a bright base layer.
This level of intensity isn’t just about visual flair—it’s about reach.
Magnesium-powered fireworks can be seen clearly from far away, making them perfect for both professional pyrotechnic shows and high-quality consumer displays that leave a lasting impression.
Other Uses of Magnesium in Pyrotechnics
Beyond traditional aerial fireworks, magnesium serves other exciting roles in pyrotechnics, enhancing even smaller-scale celebrations.
Magnesium in Sparklers
Sparklers, beloved by families everywhere, frequently use magnesium. It creates those bright sparks that safely delight kids and adults alike.
This makes magnesium indispensable in family-friendly fireworks products.
Safety Considerations of Magnesium in Fireworks
While magnesium makes fireworks spectacular, it also demands careful handling to ensure safety for everyone involved.
Handling and Storage of Magnesium
Magnesium is highly reactive and requires safe storage practices. At Red Apple Fireworks, we prioritize safety, ensuring magnesium-infused fireworks meet strict standards.
Always store fireworks in cool, dry areas away from ignition sources and follow recommended guidelines.
Firework Safety and Magnesium’s Reactivity
Due to magnesium’s intense burning properties, fireworks containing magnesium should always be handled carefully.
Responsible ignition, clear safety guidelines, and proper disposal methods protect DIY enthusiasts, families, and professionals, allowing everyone to enjoy spectacular shows safely.
Conclusion
So, is magnesium used in fireworks? Absolutely! Magnesium delivers brilliant, unmatched white sparks, transforming fireworks displays into breathtaking spectacles.
At Red Apple Fireworks, we're committed to providing safe, vibrant, and memorable fireworks for DIY enthusiasts, families, event planners, and budget-conscious buyers alike.
From our missiles fireworks to our novelty fireworks collections, we’ve got everything you need to elevate your next holiday or special event.
Looking for unbeatable deals? Join our Club Red Apple® membership and save up to 25% on your next purchase!
When you become a member, you unlock exclusive perks and benefits that make every firework show even better.
Ready to make your next event unforgettable? Visit Red Apple Fireworks today and let us help you light up the sky!
If you want to learn more, check out these articles below
- How Do Fireworks Work
- Can You Do Fireworks in the Rain?
- How Long Do Fireworks Last?
- What Causes Fireworks?
- What Exactly Is a Firework?
- How Are Fireworks Made?
- Where Are Fireworks Made?
- Is There Gunpowder in Fireworks?
- How to Light Fireworks
- When Were Fireworks Invented?
- First American Fireworks
- Facts About Fireworks
- Why Do Celebrate with Fireworks?
- How Do Fireworks Get Their Color?
- What Element Makes Purple Fireworks?







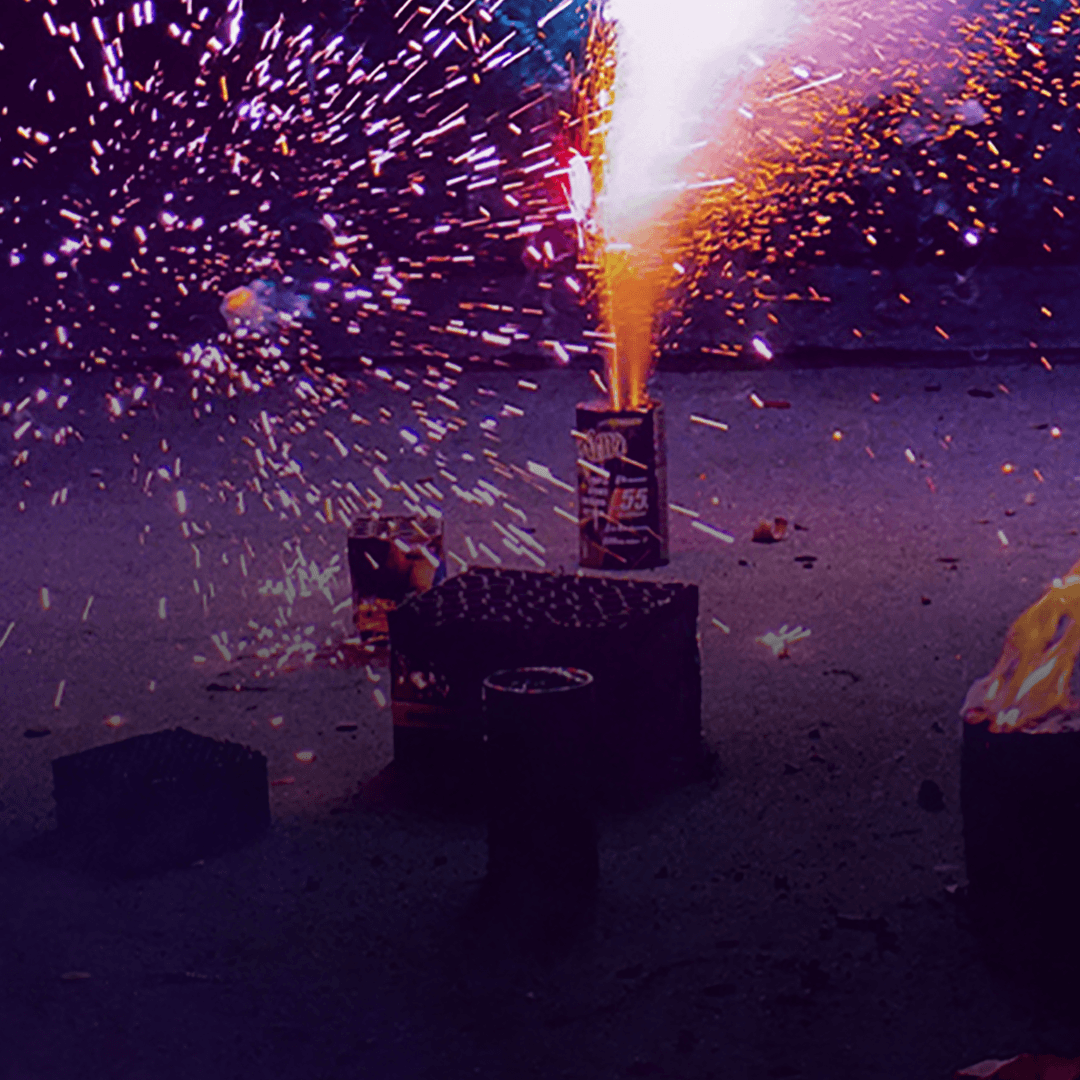




Leave a comment
All comments are moderated before being published.
This site is protected by hCaptcha and the hCaptcha Privacy Policy and Terms of Service apply.